
Cardiohelp Supplies needed: o Hardware: Cardiohelp-i on cart, O2 flow meter, two metal tubing clamps o Disposables: Maquet HIT Advanced Disposable Set o Fluids: 2 liters of Normosol-R or Plasmalyte-A. Set Up o Ensure console and all auxillary pieces are clean. O Open the guard on the Cardiohelp-i. Extremely Portable Heart-Lung Machine From Maquet. Recently on show at Arab Health 2014, the CARDIOHELP system from Maquet is the world’s smallest heart-lung machine. Weighing just 10 kg, the CARDIOHELP is very portable and can be easily carried by one person.
Abstract Objective: Cardiogenic shock is associated with mortality rates up to 70%, even if patients are treated with intensive care support or thrombolytic therapy. Early coronary revascularisation can be life-saving but it is oftentimes not available at the hospital to which the patient was initially taken. Up to now, transferring patients in a state of severe cardiogenic shock and/or cardiopulmonary resuscitation is mostly decided to be impossible.
We report on the use of two newly developed minimised systems for hand-held-extracorporeal membrane oxygenation (ECMO) (ELS-System™ and CARDIOHELP™, both from MAQUET Cardiopulmonary AG, Germany), which we have used for rapid extracorporeal life support and interhospital transfer on Mini-ECMO. We characterise the emergency use, safety instructions and bridging function of these novel Mini-ECMO devices. Methods: Between June 2007 and August 2010, we administered percutaneous Mini-ECMO support to 21 adult cardiogenic shock patients. The main components of each Mini-ECMO system are a centrifugal pump and a membrane oxygenator. The systems can act independently from wall connection points, and are they are small enough to be attached nearby a patient’s side during transport. Results: On ECMO, gas exchange improved significantly, vasopressor support could be significantly marked down and signs of shock disappeared in all patients. Emergency medical service was carried out in re-established blood-flow and oxygen delivery.
The patients were bridged to coronary artery bypass grafting (CABG) ( n = 6), ventricular assist device ( n = 1) or pulmonary embolectomy ( n = 1) or specialised intensive care ( n = 13). Thirteen patients (62%) had completely recovered. Conclusions: The use of hand-held Mini-ECMO systems enables for the first time the rapid onset of extracorporeal life support independent from the patient’s current location. However, success is extremely time- and team dependent. Highly skilled interdisciplinary patient management is essential to let minimised-ECMO become a new and highly effective bridge to recovery in out-of-centre cardiogenic shock patients.
, 1 Introduction Cardiogenic shock is caused by a severe decrease in cardiac output with subsequent tissue hypoperfusion and inadequate oxygen delivery. Rapid initiation of measures to improve cardiac output, oxygenation and ventilation are of vital importance until a patient has access to a medical facility that can perform myocardial revascularisation (e.g., percutaneous coronary interventions (PCIs), coronary artery bypass grafting (CABG), pulmonary thrombectomy or ventricular assistance (ventricular assist device, VAD)) or specialised intensive care. Even with well-managed care, cardiogenic shock patients can reach a stage at which medical therapy is insufficient to sustain an acceptable level of cardiac output. In these patients, extracorporeal life support (veno-arterial extracorporeal membrane oxygenation, ECMO) can be an effective treatment and bridging option until further cardiac treatment is available or recovery of native heart function can be established. ECMO support in adult patients with cardiopulmonary failure has been shown to improve survival, but it is rarely available, and, up to now, was limited to hospitals that have cardiac surgery departments. The use of hand-held Mini-ECMO systems, even ‘out-of-centre’, was introduced in 2008 in the medical treatment of cardiopulmonary failure, and is associated with improved survival rates in patients with refractory cardiac- and cardiopulmonary failure.
In this work, we have reviewed our data retrospectively to report on the ‘out-of-centre’ use and bridging function of two newly developed Mini-ECMO systems in adult cardiogenic shock patients. 2 Materials and methods Between June 2007 and August 2010, we performed ‘out-of-centre’ Mini-ECMO by using our first-generation Mini-ECMO device (ELS-System™, MAQUET Cardiopulmonary AG, Hechingen, Germany) in 20 cardiogenic shock patients. In one patient, we used a newly developed ultracompact Mini-ECMO device (CARDIOHELP, MAQUET Cardiopulmonary AG, Hechingen, Germany). All patients were primary located in five different outlying medical facilities (out of centre).
Both the Mini-ECMO systems we have used are suitable for easy hand-held use. Our tertiary medical-care centre preserves an interdisciplinary round-the-clock ECMO team with a field of responsibility of approximately 70.550 km 2.
The ECMO service is available 24 h day −1 and is integrated and operated by the air-medical service. Our ECMO team consisted of a consultant cardiac anaesthesiologist, a senior cardiac surgeon and a perfusionist supported by a nurse. In our patients, cardiogenic shock follows acute myocardial infarction (AMI) or severe pulmonary embolism. AMI ( n = 20) was diagnosed if a patient had significant ST-elevation in the electrocardiogram (ECG) and/or elevated cardiac enzymes (e.g., troponin I and creatinine kinase MB (CK-MB)).
Severe pulmonary embolism ( n = 1) was diagnosed by the presence of an embolus on echocardiography in patients requiring cardiopulmonary resuscitation (CPR). Cardiogenic shock was diagnosed by signs of inadequate tissue perfusion and/or hypoxaemia despite having an adequate volume status, ventilator support and vasopressor support or intra-aortic-counterpulsation (IABP). All patients had severe gas-exchange failure and required intubation and advanced ventilator support, using positive end-expiratory pressure (PEEP) and an F iO 2 (fraction of inspired oxygen) of 1.0. Indication for veno-arterial ECMO support was cardiogenic shock resistant to current available medical treatment.
Exclusion criteria were potentially lethal co-morbidity (e.g., cancer in end stage), unobserved cardiac arrest and patients in whom signs of ineffective CPR were observed. Survivors were defined as 30 days’ survival after ECMO implantation. Non-survivors died within 30 days after ECMO implantation. Percutaneous cannulation for femoro-femoral veno-arterial ECMO was carried out using Seldinger’s technique without routine use of skin incision for cannula placement.
Depending on the patient’s biometric data, we used either a 21 or 23 Fr cannula (Femoral cannula, DLP Medtronic, Minneapolis, USA or MAQUET Cardiopulmonary AG, Hechingen, Germany) for venous outflow and a 15 or 17 Fr cannula (Novaport™, Novalung, Talheim, Germany or MAQUET Cardiopulmonary AG, Hechingen, Germany) for the arterial backflow. In patients who had been heparinised or had received dual platelet inhibition, we initially performed heparin-free ECMO. In the latter course, we administered low-dose heparin to a partial thromboplastin time (PTT) that was approximately double the normal range or an activating clotting time (ACT) value above 150 s.
For veno-arterial ECMO, the tip of the arterial cannula (inflow) was positioned in the common iliac artery or the distal abdominal aorta, whereas the tip of the venous cannula (outflow) was placed in the inferior vena cava, approximately as close to the right atrium as possible to optimally decrease the preload on the failing heart. On ECMO, pump flow rates were initiated up to 4.5 l min −1 and the gas supplied to the oxygenator was initially pure oxygen with a flow rate of up to 12 l min −1 to allow for maximum gas-exchange capacity. Shortly after implementation of ECMO, the flow and gas supply rates were adjusted using blood gas examinations to meet patient’s demand. Interhospital transfer to our medical care centre was carried out after setting the patients on ECMO. Patient monitoring included invasive blood pressure measurement, ECG (electrocardiography), capnography and pulse oximetry. The arterial line was placed in the right radial artery, and blood gas measurements were drawn from that line to ensure sufficient blood flow and oxygen supply to the supra-aortic vessels. Catecholamine administration was titrated down to maintain a mean arterial pressure between 50 and 60 mmHg.
All patients were in a state of general anaesthesia and controlled ventilation modes using PEEP up to 15 cmH 2O. The patient’s body temperature was measured during transfer to prevent severe hypothermia on ECMO. According to our standard operation procedure for out-of-centre ECMO, our ECMO equipment is totally self-dependent and includes aseptic cover sheets, various cannula sizes, various guide wires and surgical equipment for vessel preparation and a hand crank for the ECMO device.
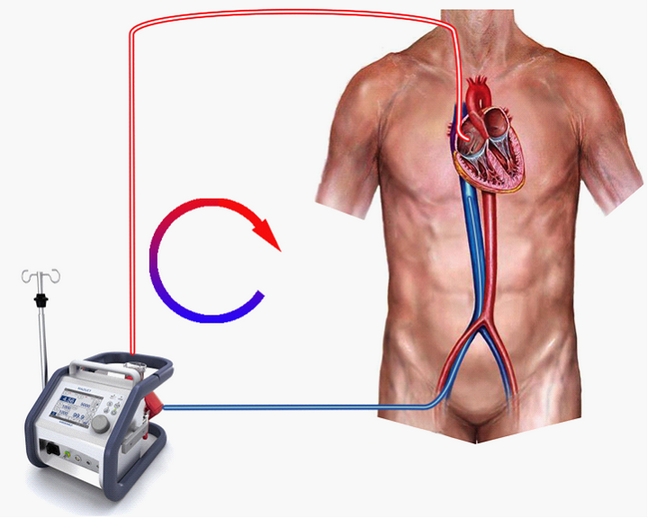
After arrival at our medical centre, we regulated the patient’s temperature to induce mild hypothermia (34 °C within 24 h) to allow cerebral protection during the patient’s post-resuscitation care. The main parts of these new Mini-ECMO devices we have used are a plasma-resistant polymethylpentene membrane oxygenator and a centrifugal pump. Both Mini-ECMO systems are suitable for easy hand-held use and fixation closed to the patient’s side ( ). The first-generation hand-held ECMO device (ELS-System™ MAQUET Cardiopulmonary AG, Hechingen, Germany) consists of a control and steering unit and a multifunctional holder system for the membrane oxygenator and the centrifugal pump. Both parts are connected with a drive line.
The total weight of the ELS-System is 24 kg. The second-generation device (CARDIOHELP™, MAQUET Cardiopulmonary AG, Hechingen, Germany) has with a total weight of 9 kg, and consists of a single hybrid pump–oxygenator unit. This newly developed system has an integrated venous probe to measure venous oxygen saturation, haemoglobin, haematocrit and venous temperature ( ). Both systems are licensed and approved for air- and ground medical transport and long-term use. The ECMO circuits are pre-connected and suitable for rapid setup and priming. Both ECMO systems are coated with heparin from tip to tip (Bioline™, MAQUET Cardiopulmonary AG, Hechingen, Germany).
The priming volume is less than 600 ml normal saline. The Mini-ECMO support during medical transfer is possible for nearly 90 min independently from wall connection points for power and oxygen supply.
After arrival at the advanced medical care centre, each cardiogenic shock patient was re-evaluated to decide on the best course of further medical treatment. Diagnostic tests, percutaneous transluminal coronary intervention (PTCI), CABG and specialised intensive care support were carried out on Mini-ECMO without the need for an ECMO system or circuit change.
Depending on the patient’s clinical course, blood coagulation was assessed as frequently as every 4 h. To recognise limb ischaemia caused by the femoral artery cannulation as soon as possible, clinical examination, pulse oximetry and ultrasound measurement were routinely performed. Hand-held Mini ECMO (CARDIOHELP, MAQUET Cardiopulmonary AG, Hechingen, Germany). 3 Results Between June 2007 and August 2010, we treated 21 adult cardiogenic shock patients with percutaneous femoro-femoral veno-arterial Mini-ECMO support.
One patient was excluded from Mini-ECMO support because signs of ineffective CPR were observed, and a further three patients were excluded in this study because CPR was terminated before the ECMO team had arrived. Cardiogenic shock followed AMI in 20 patients and severe pulmonary embolism in one patient. Percutaneous femoro-femoral veno-arterial vessel cannulation was performed successfully and uneventfully in all patients. Sufficient pump flow and gas exchange were achieved in all patients, and signs of inadequate tissue perfusion (shock) disappeared on ECMO. The median age of the survivors was 45 (21–73) years, and their median Sequential Organ Failure Assessment (SOFA) Score was 9.7 (8–11). Four patients in the survivor group underwent episodes of mechanical resuscitation before being set on veno-arterial ECMO.
Median time on ECMO was 116 h (5–168). After arrival at the medical centre, PTCI was performed in four patients and CABG (beating heart on veno-arterial ECMO) in two patients of the survivor group. One of the surviving patients was bridged on veno-arterial Mini-ECMO to left ventricular assist device (LVAD) implantation. One patient was bridged to a conventional heart-and-lung machine for pulmonary embolectomy. Six of the surviving patients were bridged on Mini-ECMO until completely native heart recovery. Median age in non-survivors was 53 (41–63) years, median SOFA Score was 11.5 (6–16) and median time on ECMO 93 h (3–166). Only pH value assessed pre-ECMO showed a significant difference between survivors and non-survivors.
Three patients in the non-survivor group received mechanical resuscitation before being placed on veno-arterial ECMO ( ). Two patients in the non-survivor group received emergency CABG procedures after arrival at the tertiary care centre. One patient died of intractable ventricular fibrillation, and five patients died of septic multiple organ failure after weaning from ECMO. Two patients died before weaning from ECMO by diagnosed brain death. In both patient groups, no technical problems occurred on ECMO, even during air- and ground medical service. Shows the gas exchange and haemodynamic parameters before and during ECMO for all patients.
On ECMO, the gas exchange improved and the oxygenation ratio (OR) increased significantly to normal range, while CO 2 value decreased significantly. Vasopressor support could be marked down significantly and the pH value increased significantly into the normal range. Non-parametric data are presented as median and minimum–maximum, and parametric data as mean ± standard deviation. Statistical tests were Wilcoxon signed-rank test, Mann–Whitney test and one-way analysis of variance (ANOVA).
For statistical analysis, we used Statistical Package for Social Sciences (SPSS) 15.0 (SPSS Inc., Chicago, IL, USA). In two patients (one survivor and one non-survivor), limb ischaemia was detected in the course on ECMO, and the arterial cannulas were switched to the right subclavian artery, and the patients underwent femoral artery reconstruction. All patients. Gas exchange and hemodynamic parameters before and during Mini-ECMO. Data are presented as median (minimum-maximum). 4 Discussion Cardiogenic shock remains the major cause of death mostly among young patients with AMI.
Despite advanced intensive care, ischaemic injury of all organs, including the heart, will become irreversible if effective circulation is not re-established. In patients who have suffered an AMI and who are in a state of cardiogenic shock and are unresponsive to high doses of inotropic agents and vasopressors and/or IABP, the initiation of extracorporeal life support can quickly restore systemic circulation and gas exchange and prevent further ischaemic tissue injury. Successful initiation of veno-arterial ECMO in severe cardiogenic shock patients reduces the need for high-dose vasopressors that have side effects and worsen myocardial and intestinal ischaemia.
Emergency percutaneous ECMO support can be highly effective in first-line treatment for various types of resistant cardiopulmonary failure. Recovery of native heart function is the primary goal in both the acute care setting and the patient’s hospital course. Reducing the time that a patient spends in cardiogenic shock and subsequent hypoxia is therefore crucial. To decrease both short- and long-term mortality rates prompt coronary revascularisation using PCIs or CABG is strongly recommended. Patients with acute chest pain are first admitted to medical facilities that are close to their homes.
These general care centres generally do not have coronary laboratories or the ability to perform cardiac surgeries (e.g., CABG, pulmonary thrombectomy or mechanical cardiac support). If prompt coronary revascularisation cannot be achieved in the general care setting through thrombolytic therapy, myocardial ischaemia will progress and cardiogenic shock becomes imminent. In these cases, advanced revascularisation strategies such as PCI and CABG are urgently required, and patient transfer in a state of severe cardiopulmonary failure becomes necessary. Even if some patients with AMIs are admitted to medical centres that can provide successful PCI, myocardial stunning can persist or post-catheterisation arrhythmias or arrests can occur. In these patients, cardiopulmonary failure also persists, much like it does in patients, who were unsuccessfully revascularised. Temporary extracorporeal assistance can therefore become an innovative strategy that can be employed until restoration of effective cardiac output can be achieved. Prior to the introduction of our Mini-ECMO system in the air medical service, patients in severe cardiogenic shock refractory to IABP support, in need of CPR and/or high dosages of vasopressor support were decided by the local physicians to be not suitable for transportation and, therefore, patient transfer was not requested.
Death of low cardiac output patients has to be accepted. Since the first hand-held Mini-ECMO system was successfully introduced in emergency medicine in 2008 by our working group, sending out specialised teams to initiate ECMO has become an innovative field in medicine. By using these newly developed hand-held Mini-ECMO systems, it is now possible to use standard air- and ground emergency medical vehicles for rapid out-of-centre extracorporeal life support. As a result, it may become possible to shorten the ‘time-to-pump time’ in cardiogenic shock patients in need of extracorporeal assistance because of the high mobility of these examined hand-held ECMO devices, which can be easily transported by standard air- and ground ambulance vehicles without time-consuming technical or personnel preplanning.
In our patients, survivors tended to be younger (median age 45 years) than non-survivors (median age 53 years). The median SOFA Score before the initiation of Mini-ECMO tended to be higher in non-survivors (11.5) than in survivors (9.7). Episodes of mechanical CPR before ECMO were necessary in four survivors and in three non-survivors, respectively. As a limitation, data for exact duration of mechanical resuscitation were not preserved in the outlying medical facilities and, therefore, could not be examined in our surviving and non-surviving patient group. Before Mini-ECMO support, non-survivors had laboratory measurements indicating that they were in more severe shock and had more prolonged tissue hypoperfusion; however, only differences in pH value were statistically significant.
These findings are in accordance with the results of a report on ECMO-assisted CPR in adults. This meta-analysis confirmed the hypothesis that patients, who had a shorter period of cardiovascular depression prior to initiation of ECMO support, would have improved survival. Because of the hand-held size and patient-side fixation of these Mini-ECMO systems, time delay and restrictions in further treatment on extracorporeal assistance is first-time avoided.
The ECMO team and the patient on Mini-ECMO can be first-time transferred using standard patient gurneys and ground- and air ambulances. PCIs can be carried out safely on Mini-EMCO. Veno-arterial ECMO provides, in patients with myocardial stunning, a bridge to native myocardial recovery and, in patients in further need of mechanical cardiac support, a bridge to VAD or heart transplantation. Although bleeding complications have often been reported in patients receiving cardiopulmonary bypass support, none of our patients experienced bleeding complications during their ECMO course, despite the presence of platelet dysfunction, anticoagulant use and co-existing coagulopathy. The tip-to-tip heparin coating of our ECMO circuits allowed most patients to be operated with minimal (5000 I.E.
Heparin) or no systemic heparinisation. Femoro-femoral veno-arterial ECMO provides both haemodynamic and gas exchange support. In practice, filling up the aorta in a retrograde manner is therefore necessary.
Creating retrograde aortic blood flow has an unknown individualised risk of supra-aortic plaque embolisation. In our patient group, we did not observe any strokes or neurological deficits. Gas exchange was restored in our patient groups rapidly and effectively within 2 h of ECMO initiation. However, veno-arterial ECMO leads to non-uniform oxygen saturation within the arterial circulation.
Even in patients with cardiogenic shock, residual myocardial function may pump blood through the lungs, which have impaired oxygenation capacity due to left-ventricular backward failure and resultant lung oedema. In this situation, the upper part of the body, including the coronary and cerebral vessels, receives poorly oxygenated blood in addition to highly oxygenated blood from the femoral artery inflow cannula. Therefore, proper patient monitoring is crucial. We recommend blood–gas-exchange monitoring in the right upper extremity to detect hypoxaemia of the upper body. When hypoxaemia is detected in the upper body despite advanced ventilator settings or limb ischaemia occurs, surgical switch of the arterial cannula from the femoral to a central site (e.g., right subclavian artery) could solve this problem. Despite potential risks, the use of hand-held Mini-ECMO systems enables, for the first time, the rapid onset of extracorporeal life support independent of the patient’s current location.
This novel technique may enable the use of ECMO in cardiac emergencies, and can be a safe and highly effective bridge to recovery for cardiogenic shock patients. Rapid-onset ECMO has become possible even for patients primary located in outlying medical facilities, and further hypoxic damage to these patients’ myocardial, systemic and brain tissues can be prevented. However, this novel technique has to be carefully introduced in pre-existing treatment strategies and in the chain of rescue even in out-of-centre CPR strategies. Proper interdisciplinary patient management is crucial to improve outcome in cardiogenic shock patients.
